:max_bytes(150000):strip_icc()/PeriodicTablecolor-58b5d9105f9b586046df7d8a.jpg)
Modern Periodic Tables recognize 118 elements.Ītoms of alike or different elements will combine to form molecules, a group of atoms, in chemical reactions. Using technology, however, scientists have been able to create additional elements that do not naturally occur. Roughly 90 of these elements occur naturally on Earth. You won’t see these variations listed on the Periodic Table, but the atomic mass that you see for various elements hints at these isotopes because it is an average of the most common masses for that element in nature.Īn element of any substance cannot be broken down into simpler substances by ordinary chemical processes. Atoms of the same element that have a different amount of neutrons than what is most common in nature are called isotopes. Usually, the number of neutrons in an element’s atom will be the same, but sometimes there are differences. Atoms of the same element will have the same number of protons in their nucleus. That means all of the elements that are alike will have atoms with the same structure and characteristics. On the next page, we will discuss in detail what each of these things mean.Īn element is a pure substance that is made up of only one kind of atom. These things help you tell the different elements apart from each other. Note that the periodic table above has a slightly different key, and the atomic mass is on top, next to the atomic number. This diagram shows you the key details you will need to identify when looking at an element on the Periodic Table: its atomic number, chemical symbol, chemical name, atomic mass, and some other details you won’t need to know immediately. :max_bytes(150000):strip_icc()/PeriodicTablenamescolor-58b5d91e3df78cdcd8d03326.jpg)
You will see that some periodic tables will give you much more information, but, for now, we will stick to the basics. The information in this box is a little simplified. It presents an enlarged version of one of the boxes you will see on the Periodic Table. When you learn more about how the Periodic Table is organized, though, you will see that it shows you a lot of useful information about elements. At first, it seems like there’s no telling what it could all mean. It’s filled with a bunch of numbers and symbols. 
– Visit iupac.Reading the Periodic Table can seem to be very confusing at first. Practical applications of isotopic measurements and technologies are included for the following fields: forensic science, geochronology, Earth-system sciences, environmental science, and human health sciences, including medical diagnosis and treatment. The background color scheme of cells categorizes the 118 elements into four groups: (1) white indicates the element has no standard atomic weight, (2) blue indicates the element has only one isotope that is used to determine its standard atomic weight, which is given as a single value with an uncertainty, (3) yellow indicates the element has two or more isotopes that are used to determine its standard atomic weight, which is given as a single value with an uncertainty, and (4) pink indicates the element has a well-documented variation in its atomic weight, and the standard atomic weight is expressed as an interval.Īn element-by-element review accompanies the IPTEI and includes a chart of all known stable and radioactive isotopes for each element. Color-coded pie charts in each element cell display the stable isotopes and the relatively long-lived radioactive isotopes having characteristic terrestrial isotopic compositions that determine the standard atomic weight of each element. The IPTEI is intended to hang on the walls of chemistry laboratories and classrooms.Įach cell of the IPTEI provides the chemical name, symbol, atomic number, and standard atomic weight of an element. 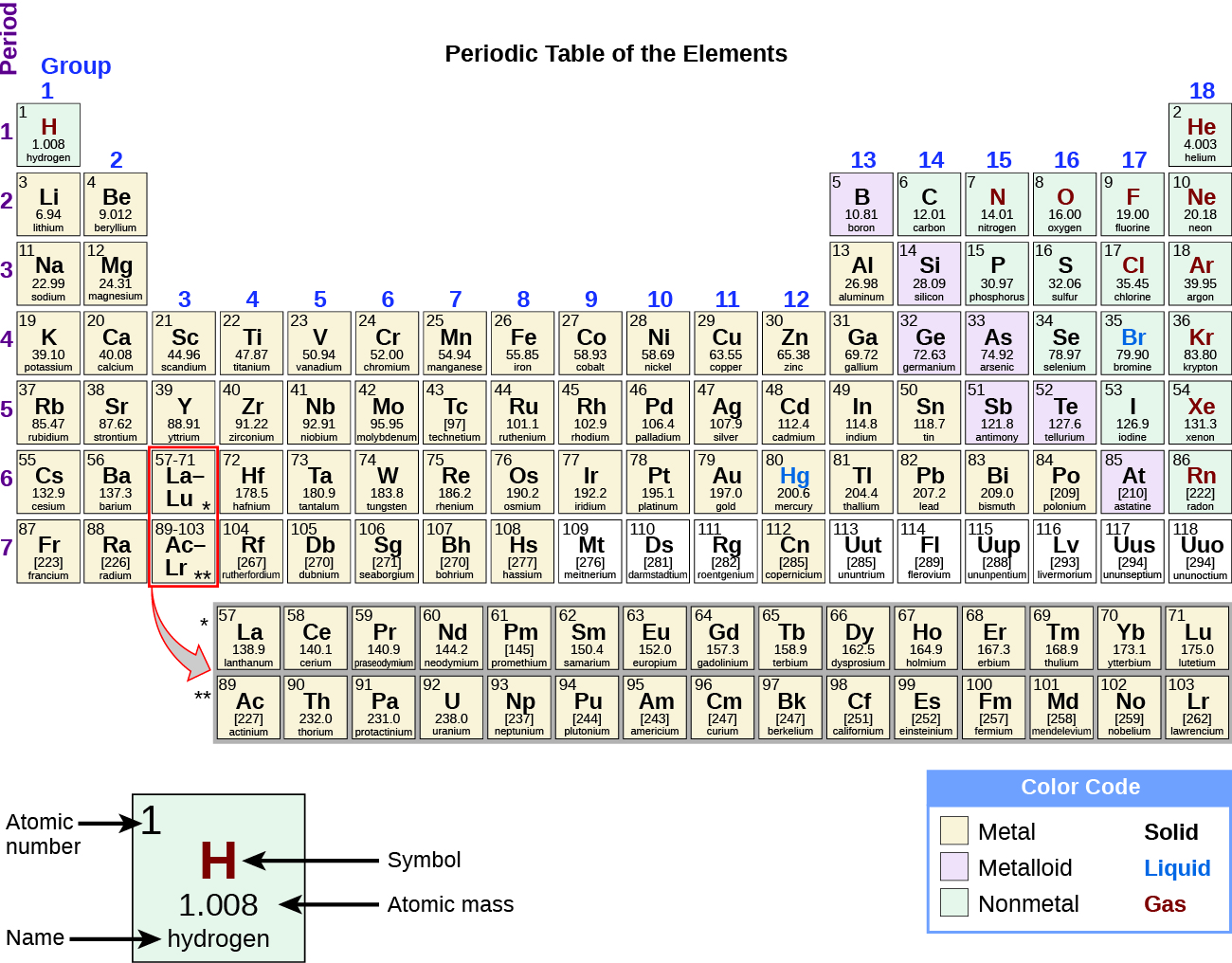
The IPTEI is modeled on the familiar Periodic Table of the Chemical Elements. eTOC Alert ‘Pure and Applied Chemistry’ – November 2023 Ībstract: The IUPAC Periodic Table of the Elements and Isotopes (IPTEI) was created to familiarize students, teachers, and non-professionals with the existence and importance of isotopes of the chemical elements.eTOC Alert ‘Pure and Applied Chemistry’ – December 2023.Green Chemistry for Life grants presented to Top Young Scientists.Teaching ethics and core values in chemistry education – Call for papers.IUPAC Green Book – New Abridged Version.Nominations Open for the 9th Polymer International-IUPAC Award.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
